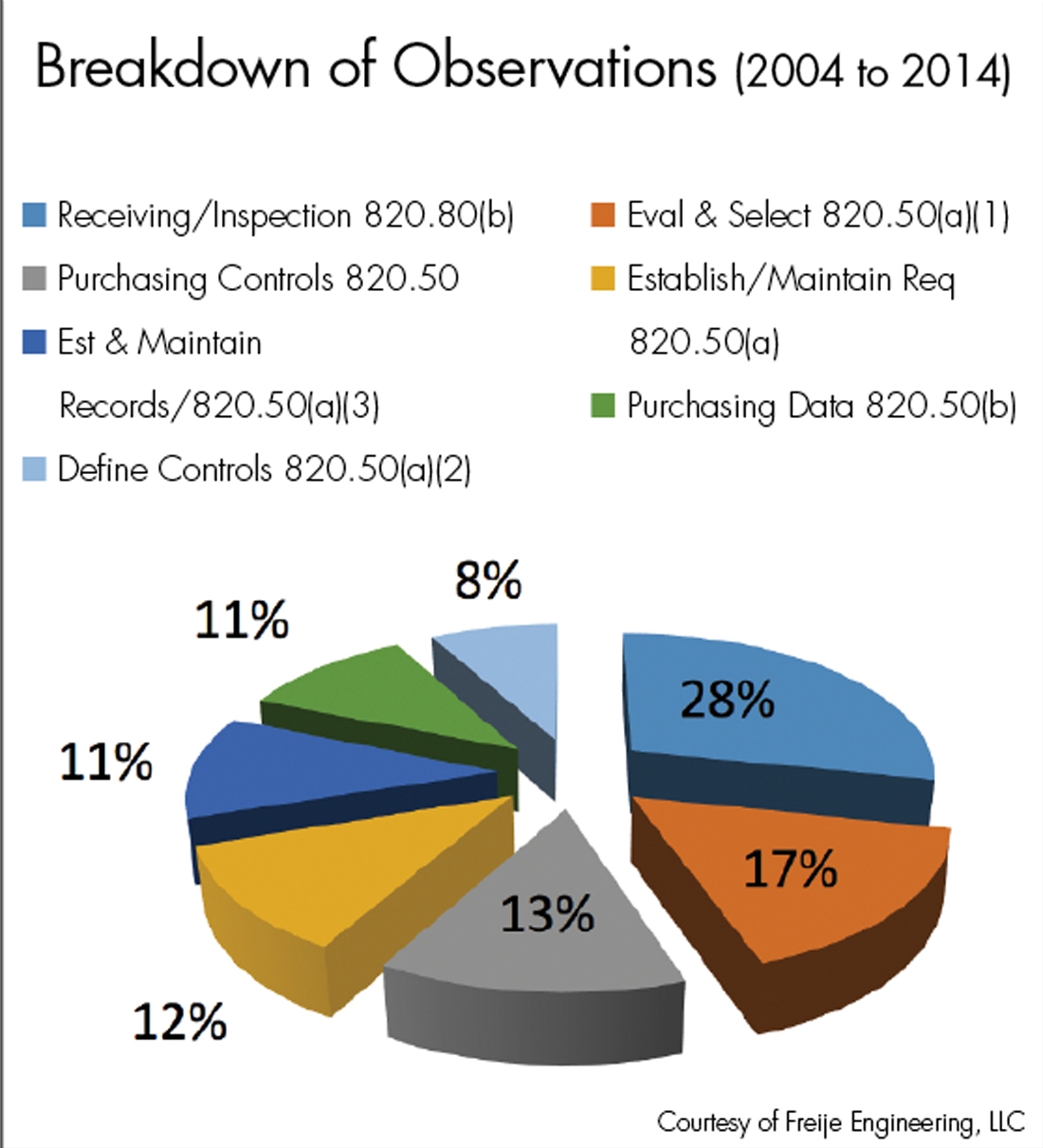
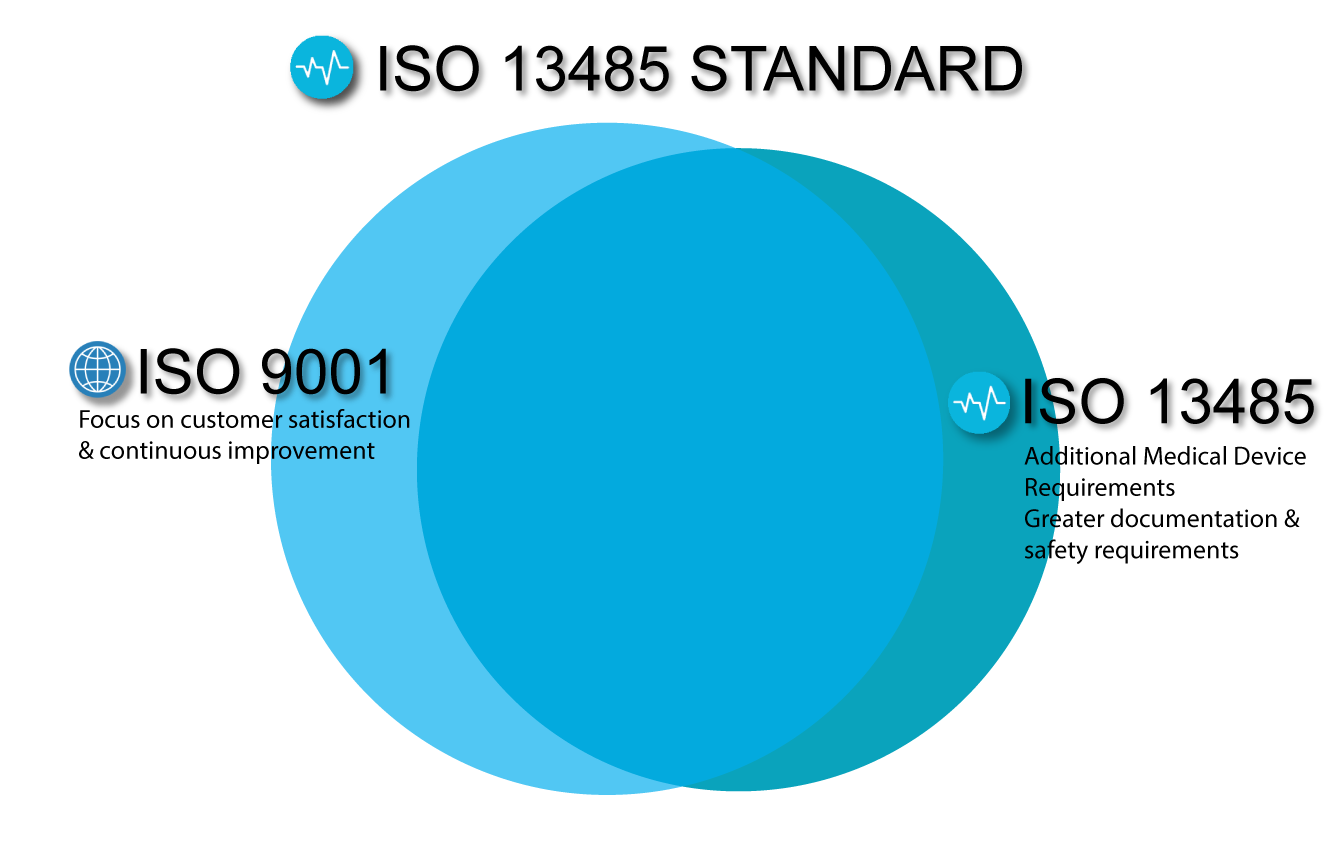
MEAN WELL acquired EN ISO-13485: 2012 Medical Devices- Quality Management System -MEAN WELL Switching Power Supply Manufacturer

DIN EN ISO 13485:2012 - Medical devices - Quality management systems - Requirements for regulatory purposes (ISO 13485:2003 + Cor. 1:2009); German version EN ISO 13485:2012 + AC:2012 (Foreign Standard)

non woven gauze|non-woven ball|Hefei Jindi Medical nonwoven fabric sheet| medical gauze|Non-woven fabrics| gauze|cotton bandage|non woven triangular bandage|non-woven triangular bandage bandage|disposable medical protective masks|protective masks ...